PESTS AND DISEASES OF FORESTRY IN NEW ZEALAND
Cypress canker
Scion is the leading provider of forest-related knowledge in New Zealand
Formerly known as the Forest Research Institute, Scion has been a leader in research relating to forest health for over 50 years. The Rotorua-based Crown Research Institute continues to provide science that will protect all forests from damage caused by insect pests, pathogens and weeds. The information presented below arises from these research activities.
Forest Pathology in New Zealand No. 8
Based on H. van der Werff (1984)
Revised by IA Hood (2007)
Causal organisms
Seiridium cupressi (Guba) Boesewinkel (Lepteutypa cupressi (Nattrass, C. Booth, & B. Sutton) H.J. Swart; in this country previously called Seiridium unicorne (Cooke & Ellis) B. Sutton = Monochaetia unicornis (Cooke & Ellis) Saccardo).
Seiridium cardinale (W.W. Wagener) B. Sutton & I.A.S. Gibson (= Coryneum cardinale W.W. Wagener).
Type of injury
Cankers form on stems, branches and in branch axils, causing dieback of leading and lateral shoots (Figs. 1 and 2). Very small trees may be killed from infection low on the stem. On older trees, stems with large cankers are prone to malformation and breakage in high winds, and death may eventually result from the combined effects of many branch cankers.

Fig.1 – A shelterbelt of Chamaecyparis lawsoniana infected with cypress canker.

Fig 2 – Severe disease in a young Cupressus macrocarpa woodlot.
Diagnostic features
- Dark brown or purple patches on bark in early stages of infection.
- Tiny, pin-head size, round, black, fruit bodies visible on the bark surface, which is often cracked (Fig. 3).
- Cankers (sunken areas) appear on branches and stem and in branch axils and may become swollen and distorted as surrounding healthy tissue continues to grow (Figs. 4, 5).
- Uneven branch and stem dieback.
- Discrete zones of yellowing and then browning distributed irregularly across the crown.
- Profuse resin bleeding.

Fig. 3 – Fruiting bodies appear as tiny, black pin-heads on the bark

Fig. 4 – Branch canker showing swelling, purple discolouration, cracked bark and resin bleeding

Fig 5. Stem canker showing cracked bark and resin bleeding.
Hosts
Chamaecyparis lawsoniana‡, Cryptomeria japonica*, × Cupressocyparis leylandii‡, × Cupressocyparis ovensii†, Cupressus arizonica†, Cupressus goveniana†, Cupressus lusitanica†, Cupressus macrocarpa§, Cupressus sempervirens‡, Cupressus torulosa†, Calocedrus decurrens†, Juniperus communis*, Sequoiadendron giganteum*, Thuja occidentalis†, Thuja plicata†.
*Rare occurrence with negligible damage.
†Low susceptibility.
‡Moderate susceptibility (for C. sempervirens, based on overseas reports).
§Very susceptible, especially in young plantations less than 10 years old.
Distribution
Cypress canker and its causal fungi are found throughout New Zealand (Fig. 6). The incidence and impact of the disease intensify as the mean temperature increases, making it more prominent in Northland and Auckland.
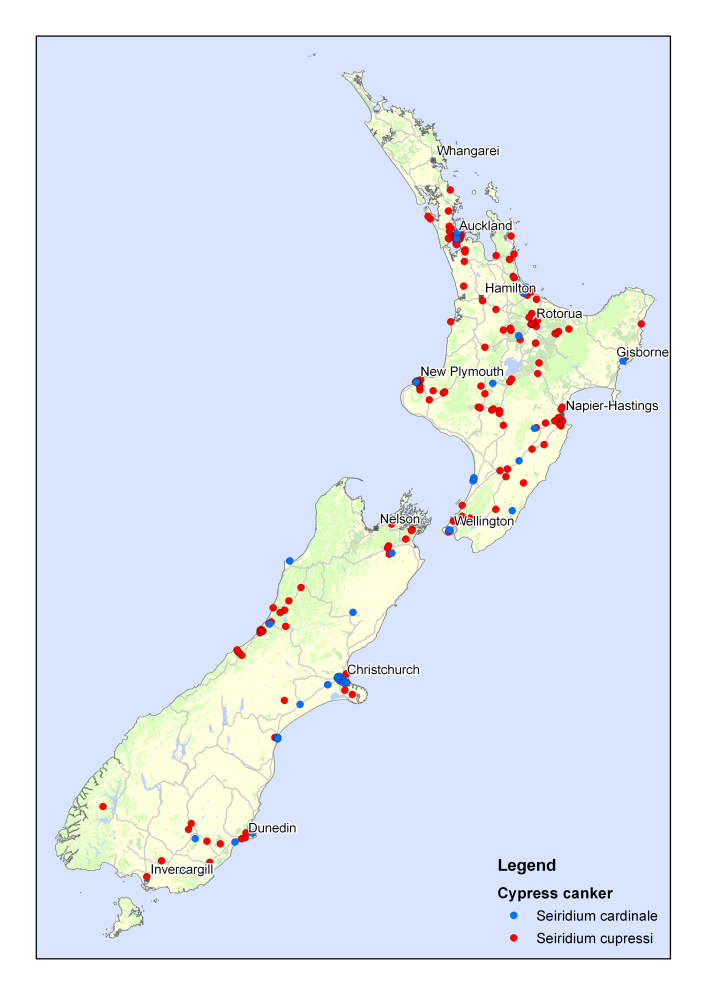
Fig. 6 – Distribution of the cypress canker fungi in New Zealand.
Disease development
Asexual spores are produced in small, black fruiting bodies which erupt through the bark surface at any time of the year when conditions are moist and warm. Spores are washed down the stem and branches or splashed from tree to tree by rain. New infections are initiated when spores lodge in bark cracks, wounds, and branch axils, and direct entry through softer undamaged tissues may be possible. Pruning stubs may also become infected, if spores are transferred from infected trees on pruning tools. Spread by these means appears to be slow and localised. The main method of dispersal over long distances seems to be through the transport of infected seed, cuttings or nursery stock, and for Seiridium cupressi possibly by spores of the sexual stage, Lepteutypa cupressi, which are thought to be wind dispersed. A sexual stage for S. cardinale is not known in New Zealand. Infected branches develop the symptoms described above, and eventually die back due to the damage caused by the cankers, and apparently also from a toxin produced by the fungi.
Economic importance
The results from two country-wide surveys indicate that cypress canker is widely distributed but uneven in impact in shelterbelts and smaller rural woodlots. The incidence of branch and stem dieback may be high in stands of Cupressus macrocarpa, Chamaecyparis lawsoniana, and sometimes × Cupressocyparis leylandii (e.g. cultivar Leighton Green), especially in warmer areas such as Auckland and Northland, but also in other localities. Infection may kill or severely malform young trees of C. macrocarpa, or reduce returns from wood production through the development of perennial stem cankers. Ch. lawsoniana is also commonly infected and the crowns in many older shelterbelts appear dishevelled as a result of irregular branch dieback. Impact is usually minor or non-existent in stands of Cupressus lusitanica, but may be significant if trees are exposed to spores from nearby diseased stands of the more susceptible species. Little cypress canker has been found on the hybrid × Cupressocyparis ovensii but its susceptibility has yet to be fully tested.
Cypress canker appears to have low impact in most large plantations, but may still be important in some stands.
Cypress canker can also be damaging to cypress cultivars used as ornamentals in urban parks and home gardens.
Control
When establishing new cypress plantations or shelterbelts in higher disease risk areas, consideration should be given to selecting a species with low susceptibility, such as C. lusitanica. Choosing even moderately resistant plants may be sufficient to lower disease levels by reducing the quantity of spores dispersing to nearby healthy trees. If planting with a more vulnerable cypress like C. macrocarpa on sites with little or no history of disease, avoid using seedlings or cuttings known to originate from heavily diseased source material. Planting stock should be disease free and not sited near to older shelterbelts or woodlots where cypress canker is present. Anecdotal observations suggest that cooler, south facing slopes discourage the disease and that stress caused by planting on exposed sites is undesirable.
If cypress canker appears, diseased branches or trees should be removed and where practical burnt as soon as they are detected, to prevent the spread of spores to other trees. Early attention will reduce the chances of a potential epidemic disease build up within a stand. In larger plantations or woodlots, especially in greater risk areas, such a labour-intensive procedure may not be feasible or successful. Even so, the felling of diseased trees during routine silvicultural thinning may still enable a healthy residual crop to be brought through to an economic harvest. Fungicidal spraying has been recommended but its effectiveness has not been tested operationally in plantations in this country, and the cost may not be justifiable. Similarly it remains to be seen whether or not wiping tools with an antiseptic during pruning prevents the spread of infection to healthy trees, though it may be a wise precaution in smaller diseased stands. Where cypress canker is present, stress from excessive pruning should be avoided, especially during dry conditions, and animal stock likely to cause damage and potentially spread spores by contact should be fenced out.
Nurseries should endeavour to supply plants that are disease free. Seed or cuttings should be taken from parent trees or stool beds that have no infection. They should not be lined out near cypress shelterbelts in which there are signs of disease. Physical damage to plants should be minimised during cultivation, and any seedlings suspected to have cypress canker should be removed. Good control has been reported overseas by spraying monthly from October to March with copper oxychloride (4 kg 50% wettable powder in 1000 litres water and 1% surfactant). A research programme is underway at Scion, Rotorua, in the hope that it may eventually be possible to provide genetic stock of C. macrocarpa showing reduced disease susceptibility.
Bibliography
Gadgil, P.D. 2005: Fungi on trees and shrubs in New Zealand. Fungi of New Zealand Volume 4. Fungal Diversity Research Series 16: 1-437.
Hood, I.A.; Gardner, J.F.; Kimberley, M.O.; Gatenby, S.J.; Cox, J.C. 2001: A survey of cypress canker disease. New Zealand Tree Grower 22: 38-41.
Self, N.M.; Chou, C.K.S. 1994: Pruning effect on incidence and severity of Seiridium cypress canker in a stand of Cupressus lusitanica. New Zealand Journal of Forestry Science 24: 75-77.
Van der Werff, H. 1984: Cypress canker in New Zealand plantations. New Zealand Journal of Forestry Science 18: 101-108.
Van der Werff, H. 1984: Cypress canker. New Zealand Forest Service, Forest Pathology in New Zealand No. 8.
This information is intended for general interest only. It is not intended to be a substitute for specific specialist advice on any matter and should not be relied on for that purpose. Scion will not be liable for any direct, indirect, incidental, special, consequential or exemplary damages, loss of profits, or any other intangible losses that result from using the information provided on this site.
(Scion is the trading name of the New Zealand Forest Research Institute Limited.)

 Farm Forestry New Zealand
Farm Forestry New Zealand

