PESTS AND DISEASES OF FORESTRY IN NEW ZEALAND
Update on SFF Paropsis biocontrol project May 2016
May, 2016.The Tasmanian parasitoid wasp, Eadya paropsidis, is being investigated as a potential biological control agent for Paropsis charybdis, the Eucalyptus tortoise beetle. The first year of the three year Sustainable Farming Fund (SFF407964) and industry co-funded project, led by the Farm Forestry Association, has been a busy one.
Laboratory Rearing in NZ
Laboratory rearing of the braconid parasitoid Eadya paropsidis in Scion’s high security containment facility has continued to challenge scientists working on this project. Some adult parasitoids did emerge successfully between mid Nov 2015 and Jan 2016. This was exactly when expected and proved that the over-wintering population is viable.

The first ever observation of the mating of a virgin Eadya paropsidis female (Fig 1) was made in December 2015 and captured on video. (The male was reared from Moina forest and the female from Runnymede forest in Tasmania; both were offspring of the 2014 importation.) Mating is important as it ensures fertilised eggs will produce both male and female offspring in the next generation.
Despite these exciting breakthroughs, improvements are needed in the parasitoids’ ability to spin complete cocoons and emerge successfully after over-wintering in the lab-reared colony. The most frustrating aspect is that when the parasitoid larvae burst out of the remains of their P. charybdis larval host, they sometimes spin incomplete cocoons. These silken pupae often contain a “window” that seems to allow the pupating wasp to dry out and die.
To complete the experiments planned for the 2015/16 summer, lab-reared E. paropsidis adults were supplemented with a fresh importation of 75 adult females collected by Dean Satchell with hand-nets from Runnymede forest (a Eucalyptus nitens plantation), north of Hobart, Tasmania. Once the parasitoids imported in mid-December had “settled in” to the laboratory cages (it took about 48 hours for their behaviour to appear normal) they were used for host specificity testing and mass-rearing of the next generation.
Two summer students were employed at Scion in Rotorua, and one honours student at UTAS in Hobart, to assist with the busy summer of laboratory and field work. Both Rotorua students have completed detailed reports on the results of attempted mass-rearing, and the non-target beetle species biology, respectively. Highlights of their work are reported below. Final results will be obtained from the honours student in Hobart later in 2016.
Host specificity testing at Scion
Host specificity testing (sometimes known as safety testing) is a very important component of the risk assessment that will ultimately be undertaken by the EPA (Environmental Protection Authority) to determine if E. paropsidis is safe enough to introduce into the New Zealand environment. Great progress was made towards this goal this summer.
The three non-target species tested this summer were:
- Tutsan leaf beetle, Chrysolina abchasica, which is currently under consideration by the EPA for release in New Zealand against the serious weed Tutsan. http://www.landcareresearch.co.nz/science/plants-animals-fungi/plants/weeds/biocontrol/research/projects/tutsan.
- Broom leaf beetle, Gonioctena olivacea, a biological control agent for the environmental weed Scotch broom http://www.landcareresearch.co.nz/science/plants-animals-fungi/plants/weeds/biocontrol/research/projects/broom
- Heather beetle, Lochmaea suturalis, a biological control agent for the environmental weed Heather http://www.landcareresearch.co.nz/science/plants-animals-fungi/plants/weeds/biocontrol/research/projects/heather

Testing methods involved:
- No-choice tests with larvae that were exposed to female parasitoids for 24 hours and reared to see if they were attacked or supported any parasitoid development.
- No-choice sequential behavioural observation tests to see if the parasitoid finds non-target larvae more or less stimulating to attack than P. charybdis larvae.
- Two-choice behavioural observation tests (Fig 2) where P. charybdis and another species of larva are presented to the parasitoid at the same time.
Tables 1 and 2 summarise the behavioural testing that has been conducted to date:
| Non-target | No-choice sequential | Two-choice behavioural | ||
| Proportion attacked Paropsis | Proportion attacked non-target | Proportion attacked Paropsis | Proportion attacked non-target | |
| Tutsan leaf beetle | 14/15 | 2/15 | 15/16 | 3/16 |
| Broom leaf beetle | 9/13 | 1/13 | 16/16 | 0/16 |
| Heather beetle | 12/12 | 1/12 | 12/12 | 0/12 |
| Non-target | No-choice sequential | Two-choice behavioural | ||
| Mean attack Paropsis | Mean attack non-target | Mean attack Paropsis | Mean attack non-target | |
| Tutsan leaf beetle | 3.8 | 0.1 | 2.9 | 0.15 |
| Broom leaf beetle | 3.4 | 0.2 | 2.7 | 0.0 |
| Heather beetle | 5.7 | 0.3 | 3.9 | 0.0 |
It appears that Eadya paropsidis females are reluctant to attack any of the non-target species larvae that were tested, especially when compared to the number of attacks on P. charybdis. Often, the parasitoids appeared to be searching the non-target foliage and walked over the non-target larvae, not even recognising that they were there. Very occasionally, they would notice them and only a few attacks were observed. However, Eadya was never reared from any attacked non-target larvae. These data are now being analysed to quantify the observed trends.
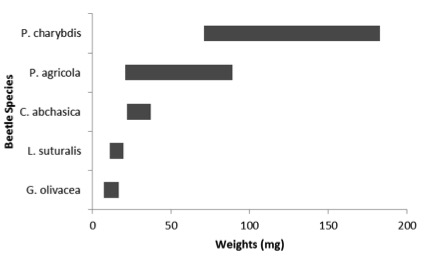
The biology of all three non-target species reared this summer was studied. The non-target larval development time and pre-pupal weight (the stage that Eadya emerges from when its development is complete) were measured and may be too small to support full Eadya development. The only possible exception to this was the Tutsan beetle pre-pupae, which are similar in size to the smallest paropsine hosts in Australia (e.g. Paropsisterna agricola). The relative sizes of the larvae are shown in Figure 3.
Scion is planning to host test at least two more non-target species in the summer of 2016/17 and will need another live importation of adult female E. paropsidis to help ensure that this is successful.
Field biology and rearing experiments in Tasmania
Our collaborators at the University of Tasmania have also had a busy summer collecting E. paropsidis from the field during November and December and studying them to increase our understanding of this parasitoid in its natural environment. Dawn and daytime collections of adults on the wing found the sex ratio was 50:50. However, dusk collections were almost 100% female. Flight activity will be analysed by time of day, temperature, light and wind conditions.
Females held individually in the laboratory at 15 °C and 16:8 L:D lived between 3 and 44 days after field collection (average 19 days). Mass-rearing from those female parasitoids produced over 200 offspring in cocoons. Experiments were undertaken to manipulate the photoperiod experienced during larval rearing and cocoon spinning. Further manipulation of photoperiod and temperature is underway during pupal development to see if laboratory rearing methods can be improved. The outcome of these experiments will be available later this year. Finally, to complete our knowledge about E. paropsidis in Tasmania, molecular identification of hosts and wasps from Paropsis charybdis, Paropsis tasmanica, Paropsisterna agricola, Paropsisterna bimaculata, Paropsisterna aegrota, Paropsisterna variicollis and Paropsisterna selmani are being carried out in North America.
Contact
Specific questions or clarification can be obtained by contacting the Project Lead and Scion entomologist, Dr Toni Withers: toni.withers@scionresearch.com



