PESTS AND DISEASES OF FORESTRY IN NEW ZEALAND
Golden-haired Bark Beetle Hylurgis ligniperda
Scion is the leading provider of forest-related knowledge in New Zealand
Formerly known as the Forest Research Institute, Scion has been a leader in research relating to forest health for over 50 years. The Rotorua-based Crown Research Institute continues to provide science that will protect all forests from damage caused by insect pests, pathogens and weeds. The information presented below arises from these research activities.
Forest and Timber Insects in New Zealand No. 18
Golden-haired Bark Beetle.
Revised 2009 (J. Bain, L.A. Berndt, B. Gresham)
Based on J. Bain (1977)
Insect: Hylurgus ligniperda (F.) (Coleoptera: Scolytidae)


Fig. 1a & b - Hylurgus ligniperda adult. The line shows natural length.
Type of Damage
Hylurgus ligniperda breeds in the inner bark of recently felled logs (especially those in contact with the ground), in stumps, and in the roots and bases of dead trees. Neither adult nor larval tunnels engrave the sapwood except in very small-diameter material. Reddish frass (bore dust) is expelled from the adult tunnels and is found throughout the workings in the inner bark.
Hosts
This beetle is apparently restricted to Pinus (pine). In New Zealand it is found mainly in Pinus radiata.
Distribution
Hylurgus ligniperda is widely distributed in Europe, the Mediterranean area, and Atlantic islands and is established as an introduced species in Japan, parts of South Africa and North and South America, Sri Lanka, and southern Australia. It was first discovered in New Zealand in 1974 near Whitford, south of Auckland.
Economic Importance
Hylurgus ligniperda breeds in logging wastes, stumps, roots and logs of harvested pine species (Reay & Walsh 2001). The damage to the wood is unimportant, except perhaps for its association with a minor degree of sapstain in the outermost sapwood of logs. However, its presence in export logs is a market access issue, and requires treatment under the International Plant Protection Convention 1997. Infested logs must either be refused for export, or be fumigated immediately before shipping. Currently methyl bromide is used for fumigation, but this is likely to be phased out in the near future (Pawson et al 2009).
Garcia de Viedma (1964) reports that in Spain pines up to 15 cm in diameter may be ringbarked and killed when, during the winter, groups of 30-40 adult Hylurgus ligniperda make communal galleries in the larger roots and the root collar. Damage of this sort has not been reported elsewhere, either overseas or New Zealand.
Description
The beetles (Fig. 1a, b) are about 6 mm long and 2 mm wide. They are black, except for the antennae and the terminal segments of the legs which are reddish-brown. Most of the surface bears yellowish hairs, which are particularly obvious on the posterior slope of the elytra (wing cases) and on the front of the head. Hylurgus ligniperda is found in the same situations as Hylastes ater and, being superficially similar in appearance, may be confused with it. Hylurgus ligniperda is larger and hairier than Hylastes ater, which is only 4-5 mm long and 1.4 mm wide, and these characteristics conveniently distinguish the two species. Both Hylastes ater and Hylurgus ligniperda may be confused with Pachycotes peregrinus. This latter is about the same length as H. ater but is more humped in profile and the pronotum appears dull, not shining as in Hylastes ater and Hylurgus ligniperda. All three beetles are shown in Fig. 3.

Fig. 3 - Three beetles which may be mistaken for each other: (A) Hylastes ater, (B) Hylurgus ligniperda, (C) Pachycotes peregrinus. Note differences in shape of rear edges of pronotum.
The fully grown larvae are white, slightly C-shaped, legless grubs with yellowish-brown head capsules. Those of Hylurgus ligniperda are about 7-8 mm long and 2 mm wide. They are larger than Hylastes ater larvae and have two, almost circular, dark protruberances on the front of the head above the jaws (Fig. 4). Smaller larvae are similar, except that the body wall is more transparent, and the reddish contents of the gut, are visible. The protruberances on the front of the head can be seen, with the aid of a hand lens, in larvae that are about half-grown.
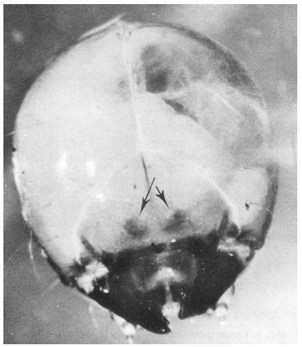
Fig. 4 - Front view of head of Hylurgus ligniperda larva. Note the two dark protruberances above the jaws.
Life History and Habits
The brood gallery, which is initiated by the female, consists of a short entry tunnel leading to an oblique nuptial chamber in the innermost bark. From this nuptial chamber the female constructs a long egg-gallery, usually parallel with the grain. The egg-gallery can be up to l m long and is not always straight (Fig. 5); it often meanders and sometimes doubles back on itself. The female is invariably found at the end of the tunnel and the male is usually found a short distance from the nuptial chamber and may assist in gallery construction by pushing out frass. Males have been observed to make short feeding tunnels branching off from the nuptial chamber or the egg-gallery. Sometimes two males have been found in the galleries, and nearly always this was before the eggs had hatched.

Fig. 5 - Larval galleries of Hylurgus ligniperda in radiata pine.
The eggs are laid in notches cut in the walls of the egg-gallery and covered with frass. They are laid over a period of at least six weeks and periods of egg-laying are interspersed with periods of feeding. Eggs are laid along approximately 100-200 mm of egg-gallery, the female will then extend the gallery for about another 200 mm and lay more eggs, and so on. As a result, the larvae tend to be found in groups - mature or nearly mature larvae near the nuptial chamber, then groups of successively smaller, even-sized larvae at intervals along the tunnel, and finally eggs just behind the female which may still be extending the gallery. The average number of eggs laid per female has not been determined. Eggs laid over the summer months hatch in about two weeks.
The larval galleries are initially at right angles to the egg-gallery but they soon become random so that they make no distinctive pattern. There are apparently four larval stages and when the larvae are fully grown they pupate at the end of their tunnels. The pupal stage lasts somewhat less than two weeks. During the summer months, development from initiation of brood galleries to the first appearance of teneral (newly-formed) adults takes 10-11 weeks.
Control
Larval competition for food is probably the most important factor in limiting numbers. No parasites or predators have been recorded in New Zealand.
Green sawn timber intended for export may be protected by the addition of an insecticide to the antisapstain bath or spray. Infestations in export logs may be minimised by:
- Rapid turnover of log stockpiles in the forest;
- Selection of unshaded skid sites;
- Stacking of logs on skids rather than on the ground;
- Use of low intensity yellow lights (e.g., low pressure sodium) for general site lighting at wood processing and transitional facilities, as this could reduce the long range visual attractiveness of a site to these, and other, wood-boring and bark beetles (Pawson et al 2009).
Monitoring of H. ligniperda (and other wood-boring and bark beetles) can be conducted using Lindgren funnel traps with lures of alpha-pinene and ethanol (Brockerhoff et al 2006).
Bibliography
Bain, J. 1977: Hylurgus ligniperda (Fabricius) (Coleoptera: Scolytidae). New Zealand Forest Service, Forest and Timber Insects in New Zealand No. 18.
Brockerhoff, E.G., Jones, D.C., Kimberley, M.O., Suckling, D.M., Donaldson, T., 2006. Nationwide survey for invasive wood-boring and bark beetles (Coleoptera) using traps baited with pheromones and kairomones. Forest Ecology and Management 228: 234-240.
Emberson, R.M. 1984. Forest and timber insects. In: Scott, R.R. (ed) New Zealand Pest and Beneficial Insects. Lincoln University, Canterbury, New Zealand. P 191-204.
Garcia de Viedma, M. 1964: Hylurgus ligniperda F., plaga de las repoblaciones de pino: sintomas de suataque.
Boletin del Servicio de plagas forestales 7: 61-63 [Review of Applied Entomology (A) 54: 128-129].
Glare, T.R., Reay, S.D., Nelson, T.L., Moore, R., 2008. Beauveria caledonica is a naturally occurring pathogen of forest beetles. Mycological Research 112: 352-360.
Pawson, S.M., Watt, M.S., Brockerhoff, E.G., 2009. Using differential responses to light spectra as a monitoring and control tool for Arhopalus ferus (Coleoptera: Cerambycidae) and other exotic wood-boring pests. Journal of Economic Entomology 102: 79-85.
Reay, S.D., Walsh, P.J., 2001. Observations of the flight activity of Hylastes ater and Hylurgus ligniperda (Curculionidae: Scolytinae) in Pinus radiata forests in the central North Island, New Zealand. New Zealand Entomologist 24: 79-85.
Reay, S.D., Walsh, P.J., 2002. Relative attractiveness of some volatiles to the introduced pine bark beetles, Hylastes ater and Hylurgus ligniperda (Curculionidae: Scolytinae). New Zealand Entomologist 25: 51-56.
Reay, S., Brownbridge, M., 2007. Biological control of exotic bark beetles. New Zealand Tree Grower 28: 24.
This information is intended for general interest only. It is not intended to be a substitute for specific specialist advice on any matter and should not be relied on for that purpose. Scion will not be liable for any direct, indirect, incidental, special, consequential or exemplary damages, loss of profits, or any other intangible losses that result from using the information provided on this site.
(Scion is the trading name of the New Zealand Forest Research Institute Limited.)



