PESTS AND DISEASES OF FORESTRY IN NEW ZEALAND
Brown lace lerp hyperparasitoid found in New Zealand
From Biosecurity New Zealand Issue 68, June 2006.
A parasitic microwasp associated with brown lace lerp, a pest of some eucalypts, has been newly recorded from New Zealand. The new microwasp, Coccidoctonus psyllae, is a hyperparasitoid, or a parasitoid that uses another such wasp as its host. C. psyllae is unlikely to be a welcome addition to our fauna as its presence will probably contribute to an increase in brown lace lerp populations.
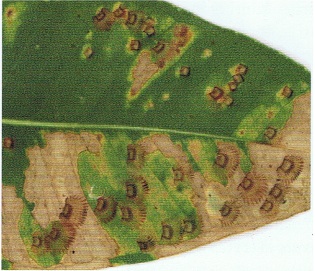
Brown lace lerp, Cardiaspina fiscella, is an Australian psyllid that attacks Eucalyptus species. Severe infestations can result in extensive damage to foliage. Brown lace lerp was first identified in New Zealand in 1996, and has spread throughout most of the North Island. It has now been recorded from a wide range of Eucalyptus species (PPIN records), but it appears that in New Zealand C. fiscella is only a significant pest of Eucalyptus botryoides, E. grandis and to a somewhat lesser extent E. saligna (D. Hocking and D. Satchell pers. comms., 2006).
Arrival of the brown lace lerp parasitoid
In late 1999, an Australian parasitic microwasp, Psyllaephagus gemitus, was recorded for the first time from New Zealand, reared from brown lace lerp in Northland (Withers 2001). At the time of its discovery, an application to import P. gemitus into containment for testing as a biocontrol agent for brown lace lerp had just been approved by ERMA. In 2000, lerp populations appeared to have decreased in Northland wherever P. gemitus was present and tree recovery was noticeable (Withers and Bain, 2000; Withers 2001).
... and hyperparasitoid
Further rearing over the summer of 2005-06 has revealed another parasitic microwasp associated with brown lace lerp in Auckland.
This species, Coccidoctonus psyllae, is a hyperparasitoid, i.e., a secondary parasitoid, or one that uses another parasitoid as a host. Species of Coccidoctonus are hyperparasitoids of various homopterans, including psyllids and scale insects, via primary hosts in the hymenopteran families Encyrtidae and Pteromalidae (Noyes 1988).

In its native Australia, C. psyllae has been reared as a common hyperparasitoid of several lerp-forming psyllids, including brown lace lerp (Riek 1962, Campbell 1992). The primary parasitoids of the psyllids recorded by these authors were "species of ... Psyllaephagus", and "P. gemitus and others" respectively. In New Zealand, C. psyllae has been reared from brown lace lerp along with Psyllaephagus gemitus. This association, along with known rearing records from Australia, indicates that C. psyllae is using P. gemitus as a primary host in New Zealand. To date, Coccidoctonus psyllae has been reared from eucalypts at three sites in urban Auckland: Mangere, Waikumete Cemetery and Mt Albert.
C. psyllae is the second species of Coccidoctonus to be recorded from New Zealand. The first, C. dubius, was deliberately introduced from Australia in 1921 for the control of black scale in the mistaken belief that it was a primary parasitoid.
Effects of hyperparasitoids on pests
In complexes involving hyperparasitoids, an inverse relationship between the rate of hyperparasitism and the level of herbivore control by primary parasitoids is often assumed. Research has shown that there is evidence for this relationship, in the short term at least. Accordingly, the presence of Coccidoctonus psyllae here is likely to reduce the level of control of brown lace lerp provided by its primary parasitoids, and high populations of C. psyllae are likely to result in higher populations of brown lace lerp. Outbreaks of two Cardiaspina species in Australian forests are thought to have been contributed to by high populations of C. psyllae and another hyperparasitoid acting to suppress Psyllaephagus gemitus, the primary lerp parasitoid (Campbell 1992).

This relationship between hyperparasitoids and herbivores has been demonstrated recently in the same ecosystem in a striking parallel: the accidental arrival of the Eucalyptus tortoise beetle hyperparasitoid, Baeoanusia albifunicle. The primary host of the hyperparasitoid Baeoanusia is the deliberately introduced tortoise beetle egg parasitoid Enoggera nassaui, which was introduced in 1987. Enoggera was initially successful in controlling its target, the tortoise beetle, but in 2001 Baeoanusia was first recorded attacking Enoggera. The hyperparasitoid has suppressed Enoggera populations so much that at times eucalypt growers have resorted to broad-spectrum insecticides to control eucalyptus beetles.
Entry pathways for parasitoids
Anecdotal evidence suggests that exotic parasitoid species may often enter New Zealand along with their hosts. New records of a number of recently introduced pest species have been followed closely by those of their respective parasitoids. This suggests that founder members of many pest species may enter the country complete with their own parasitoid complement, which may include not only parasitoids but also hyperparasitoids. Brown lace lerp was first recorded here several years before its primary parasitoid P. gemitus, which in turn was recorded before the hyperparasitoid C. psyllae. This chronology suggests repeated incursions, and the possibility that P. gemitus was deliberately introduced for brown lace lerp biocontrol has even been raised (Withers and Bain, 2000; Withers 2001). However, the evidence does not preclude the possibility that the lerp and its entire parasitoid complex entered the country simultaneously. The lower population levels and less conspicuous habit of the microwasps would result in a longer gap between establishment and recognition. Pathways suggested in the literature for entry of Australian insects include the unintentional entry of foliage associated with air cargo containers, as well as camping and golfing equipment. Eucalyptus foliage remains viable for a considerable length of time after detachment from the tree, and is capable of supporting the parasitoid complexes associated with phytophages such as lerps for several weeks, as evident during the rearing of this hyperparasitoid. Wherever possible, new phytophages should be reared in order to detect associated parasitoid and hyperparasitoid incursions.
References
Campbell, K.G. 1992: The biology and population ecology of two species of Cardiaspina (Hemiptera: Psyllidae) in plague number on Eucalyptus grandis in New South Wales. Proceedings of the Linnean Society of New South Wales 113(2) .-135-150.
Noyes, J. S. 1988: Encyrtidae (Insecta: Hymenoptera). Fauna of New Zealand 13: 1-188.
Riek, E.F. 1962: A new species of Echthroplexis, an encyrtid hyperparasite of lerp-forming psyllids on eucalypts (Hymenoptera, Chalcidoidea). Proceedings of the Linnean Society of New South Wales 87(2): 189-190.
Withers, T; Bain, J. 2000: Mysterious arrival of Cardiaspina parasitoid. New Zealand Tree Grower 21(2): 33.
Withers, T. M. 2001: Colonization of eucalypts in New Zealand by Australian insects. Austral Ecology 26(5): 467-476.
Jo Berry, Landcare Research



