Foliage sampling season begins in February
Murray Davis and Jianming Xue, New Zealand Tree Grower February 2010.
Foliage analysis is needed to determine the nutrient status of a forest crop. This analysis is to identify the cause of a nutrient disorder or poor vigour, or to confirm a diagnosis made on the basis of plant symptoms. Foliage analysis can be used to predict the need for fertiliser and assess the effectiveness and longevity of past fertiliser applications. Monitoring crop nutrients can provide an early indication of a decline.
In forestry, foliage analysis is used much more than soil analysis to work out forest nutrient status and to assess the need for fertilisers. Trees have deep root systems and are long lived, posing difficulties of where and when to take soil samples. Foliage analysis avoids these difficulties – it uses the ability of trees to exploit available nutrients over the whole soil volume occupied by the tree root system.
Sampling strategy
A number of factors affect nutrient concentrations in leaves and these must be taken into account when sampling foliage, otherwise the results may be misleading. These factors include genetic variation, seasonal variation, foliage age and crown position. It means that standard sampling procedures must be rigorously followed to ensure that analysis results can be correctly interpreted.

Samples for radiata pine are collected from mid-February to the end of March and for Douglas fir the recommended sampling time is from late March to mid-June. Foliar nutrient concentrations are generally stable over these periods.
If samples are being collected to determine fertiliser requirements, they should be taken from around 25 trees at evenly spaced distances along transects that cover the whole of the area being sampled. An alternative option is to take composite samples from fewer locations within the stand, with each composite consisting of samples taken from a cluster of five trees. However single sampling provides better coverage of the area than cluster sampling.
If sampling is to find the cause of visual symptoms or the slow growth of a patch of trees then samples from about 10 trees may be collected from trees showing the symptoms. The results can be compared with diagnostic values or with samples from healthy trees collected from the same general area.
Further points to note are −
- Samples should be collected from dominant or co-dominant trees
- Trees with heavy cone production should be avoided because of diversion of nutrients from foliage to cones
- Edge trees and trees near localised pollution sources, such as unpaved roads, limeworks or recent nearby aerial topdressing, should be avoided
- Similar amounts of foliage, a good handful, should be collected from each tree.
Sampling procedures
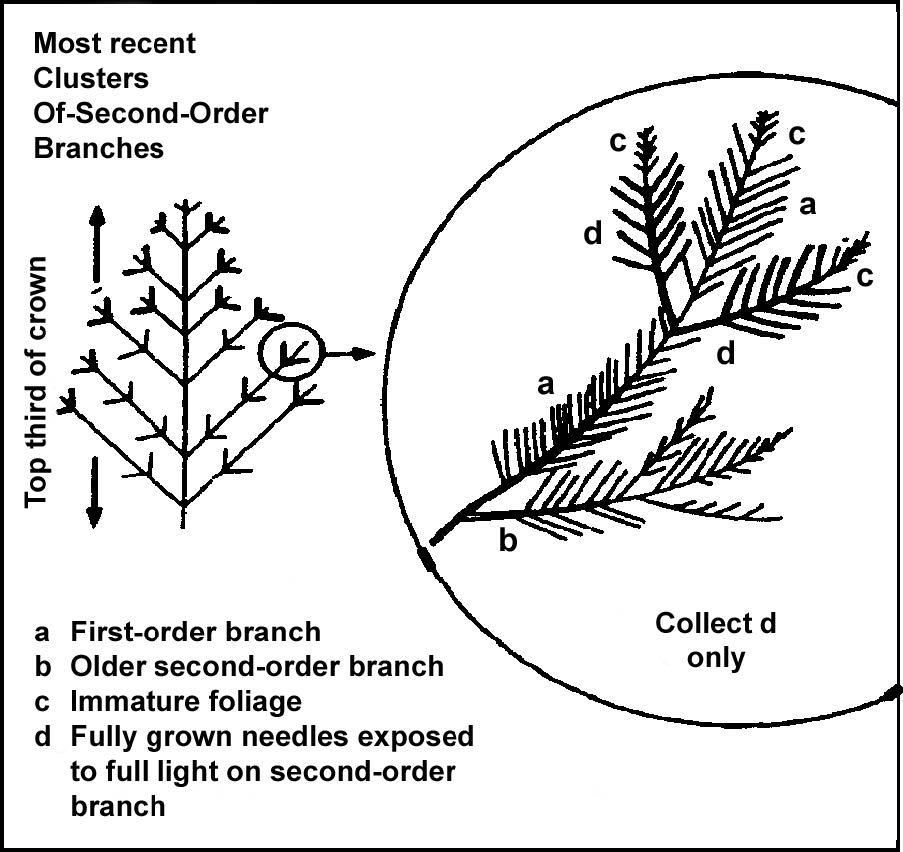
For needle leaved conifers collect samples from current season fully grown foliage on the most recent second order branches in the top third of the crown.
For eucalypts sample the first fully expanded leaves of an actively growing branch in the mid to upper canopy. The leaves should be undamaged and exposed to full sunlight. For species that have juvenile and adult foliage, sample the adult leaves.
Samples should be placed in paper or polythene bags and stored in a refrigerator, labelled and dispatched to the laboratory as soon as possible. Aim to have the laboratory receive fresh samples within 24 hours of collection or removal from a refrigerator.
Sampling tall trees
The method used to collect foliage samples is determined by tree height. For trees up to six metres in height, the samples are generally picked directly from the tree by hand. A shepherd’s crook may be useful to bring branches within reach.
For trees between six and 11 metres in height, pole mounted cutters can be used to remove small branches. Different lengths of poles can be used for stands of different heights. For trees between 11 and 30 metres a shotgun can be used to collect samples. Use a 28 to 36 gram load of No. 5 or No. 6 shot. For smaller trees you should use a No. 7 shot. Remember that ear muffs and protective glasses are essential, and for safe operation it is advisable to insert only one round.
| Deficient and adequate ranges of foliar nutrients for radiata pine and Douglas fir | ||||
|---|---|---|---|---|
| Radiata pine | Douglas fir | |||
| Deficient, Less than |
Adequate, More than |
Deficient, Less than |
Adequate, More than |
|
| Nitrogen per cent | 1.20 | 1.445 | 1.20 | 1.45 |
| Phosphorus per cent | 0.10 | 0.13 | 0.08 | 0.15 |
| Potassium per cent | 0.30 | 0.50 | 0.35 | 0.80 |
| Calcium per cent | 0.10 | 0.10 | 0.10 | 0.25 |
| Magnesium per cent | 0.055 | 0.10 | 0.06 | 0.12 |
| Sulphur per cent | - | 0.12 | 0.12 | 0.14 |
| Boron mg per kg | 8 | 12 | 12 | 15 |
| Copper mg per kg | 2 | 4 | 1 | 4 |
| Zinc mg per kg | 10 | 20 | 9 | 15 |
| Manganese mg per kg | 10 | 20 | 4 | 25 |
| Iron mg per kg | 10 | 20 | 25 | 50 |
For trees over 30 metres in height, samples can be collected by climbing as trees of this height are beyond the range of a shotgun. It is essential to use all relevant safety equipment including a safety belt which will allow use of both hands for removing samples.
Diagnostic values
Diagnostic nutrient concentrations associated with performance have been determined for many different tree species. Those for radiata pine and Douglas fir, are shown in the table. Experience with radiata pine indicates that for phosphorus, a fertiliser response is likely if concentrations fall in either the deficient or marginal ranges.
For nitrogen, potassium, magnesium, boron and copper, the values give a good prediction of fertiliser response if they are in the low, but not the marginal range. For the remaining nutrients, there is insufficient information and experience to predict a response, even in the low range. The laboratory will be able to suggest the appropriate rate and type of fertiliser to apply.

 Farm Forestry New Zealand
Farm Forestry New Zealand

